Table of Contents
Plasmapheresis is a procedure that I did not manage during my nephrology fellowship, it was run by hematology. When I entered clinical practice I found out it was my responsibility to prescribe. I don’t think this experience is unusual, plasmapheresis is often managed by hematology at academic university centers and nephrology in the community.
Since plasmapheresis involves removing blood from the body, running it through a machine and returning the purified blood to the patient, like hemodialysis, it makes sense for it to be managed by nephrology.
What is Plasmapheresis?
Plasmapheresis is the extracorporeal (outside the body) separation of plasma from blood cells. This is the most common term used, but you may also hear the terms to describe this procedure like apheresis or therapeutic plasma exchange. What’s the difference?
- Apheresis: Extracorporeal removal of a blood constituent
- Plasmapheresis: Plasma is the constituent removed from the blood. The extracorporeal removal of plasma.
- Therapeutic Plasma Exchange (TPE): Extracorporeal removal of plasma in exchange for replacement fluid
What Makes Plasmapheresis Different from Hemodialysis?
Essentially it’s which molecules are removed from blood.
Whole blood contains cells, red blood cells, white blood cells and platelets, as well as plasma. The plasma contains an aqueous phase, liquid with dissolved electrolytes, and a solid phase, solid things floating around such as proteins and lipids.
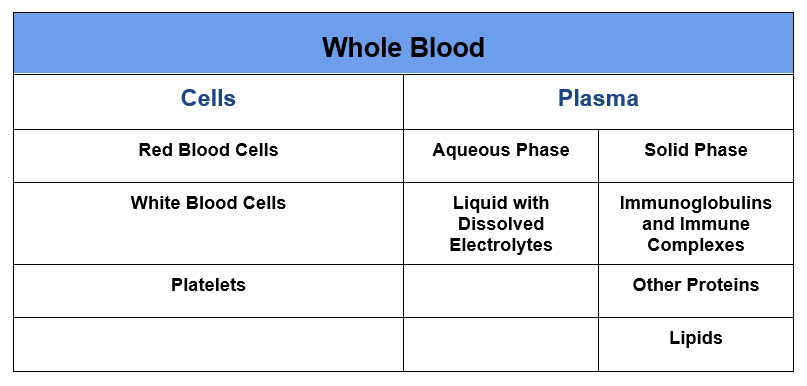
Hemodialysis removes small and middle size molecules, essentially things small enough to fit through the membrane pores, things like electrolytes, urea and creatinine. It won’t remove larger things that are too big to fit through the pores like protein, albumin, lipids or immunoglobulins or smaller molecules that are bound to proteins.
Plasmapheresis removes everything in plasma including these larger and protein bound molecules including proteins, immunoglobulins and lipids.
How Plasmapheresis Works
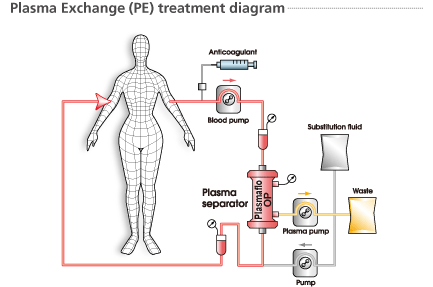
Essentially plasmapheresis takes whole blood, separates the plasma from the blood cells and discards the former.
Therapeutic plasma exchange involves discarding the separated plasma and adding a replacement fluid, typically 5% albumin or fresh frozen plasma (FFP).
The way I describe it to patients is, it takes centrifuges the blood, keeps the blood cells, gets rid of the part with the antibodies and returns the blood without these antibodies.
Although centrifuging the blood is the most common technique, it is not the only way to do plasmapheresis. There is a way to use a dialysis machine with a filter that has pores big enough to remove everything in plasma, but too small for blood cells to pass through. This is called the membrane separation technique.
Both techniques are comprised of the following steps:
- Whole blood is removed from patient
- Anticoagulant is added to the whole blood
- Plasma is separated from the blood cells
- Separated plasma is discarded
- Replacement fluid 5% albumin and/or FFP is added to replace the discarded plasma volume
- Blood with replacement fluid is returned to patient
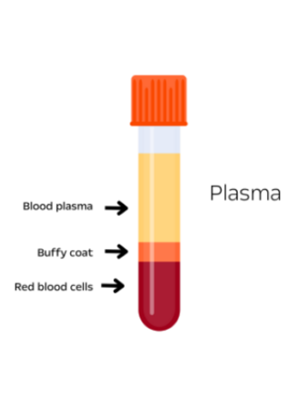
Centrifugation Technique
The most common technique used for plasma separation is centrifugation. When whole blood is centrifuged the components are separated based on density with red blood cells outermost followed by a buffy coat of white blood cells and platelets with the plasma remaining innermost.
- Specialized Plasmapheresis Machine
- Anticoagulant is typically citrate. Citrate exerts its anticoagulant effect by chelating calcium. About 80% of citrate is cleared by the pheresis, decreasing the risk of citrate toxicity
- Calcium, typically via a continuous drip, is given during the pheresis procedure to mitigate the development of hypocalcemia
- Replacement fluid is 5% albumin and/or FFP based on the clinical situation.
- More efficient allowing:
- Slower blood flow rate (10-150 ml/minute) and shorter treatment time. The slower blood flow rate allows for the possibility of using peripheral veins as opposed to a central dialysis type catheter. (Although in clinical practice a central type dialysis catheter is typically used.) Exchanging 1 plasma volume typically takes 1.5-2 hrs
Check out this video for a demonstration.
Membrane Separation Technique
- Dialysis or CRRT (Continuous Renal Replacement Therapy) machine used in hemofiltration mode.
- Specialized plasma exchange filter used in place of dialysis membrane. This filter has large membrane pores (0.3-0.5 microns), too small for cells to fit through, but large enough that any noncellular component of plasma can be removed.
- Anticoagulant is typically heparin. Only a smaller percentage of citrate is cleared by this technique, increasing the risk of citrate toxicity.
- Replacement fluid is 5% albumin and/or FFP depending on the clinical situation
- Less efficient, filtration fraction limited to 30-35%. Higher filtration fractions lead to thrombosis of the filter. Therefore, higher blood flow rates necessitating a central dialysis type catheter and longer treatment times are required. Central dialysis type catheter required. Exchanging 1 plasma volume typically takes 3 hours.
Prescribing Plasmapheresis
The prescription for plasmapheresis includes:
- Plasma volume to be exchanged
- Frequency and number of exchanges
- Type of replacement fluid
- Calcium repletion.
Plasma Volume
Typically 1.0 – 1.5 plasma volumes will be exchanged per treatment. The majority of substances in plasma are removed with this exchange volume. There is little value in doing more than 1.5 exchanges per session.
Plasma volume is calculated based on weight and hematocrit. The hematocrit (HCT) is the percentage of red blood cells (RBC) in whole blood, the majority of the remaining volume would be plasma.
Estimated Plasma Volume = (0.065 x weight kg) X (1-HCT)
For patients of extreme body weight (BMI > 35) it may be better to use 120% of ideal body weight as opposed to actual weight
Ideal Body Weight (in kg):
Males: 50 + 2.3 x (height in inches – 60)
Females: 45.5 + 2.3 x (height in inches – 60)
Ideal Body Weight and Adjusted Body Weight
Frequency and Number of Treatments
This is determined by the intravascular amount and rebound (redistribution from the extravascular to intravascular space and generation) of the pathologic substance.
Most commonly plasma exchange is performed to remove a pathologic antibody.
IgG:
- 45% intravascular
- Low turnover
- Long half life
- 6 treatments every other day will decrease IgG to < 20% of baseline
- For most conditions a course of 5-7 treatments over 2 weeks is appropriate
IgM:
- 90% intravascular
- Short half life
- Higher daily turnover
- A more significant decrease per treatment, but quicker rebound than for IgG
- Waldentroms macroglobulinemia may require daily treatments for 2-3 days
Daily plasma exchange:
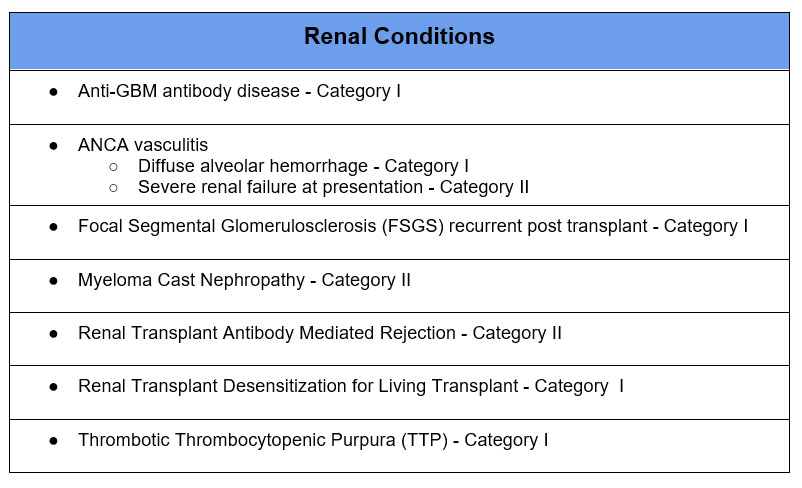
- Because of their clinical severity Thrombotic Thrombocytopenic Purpura (TTP) and anti-Glomerular Basement Membrane (GBM) antibody disease typically require daily treatments.
- TTP: Continue until platelets and LDH normal
- anti-GBM antibody disease: Continue until clinical resolution of glomerulonephritis and/or alveolar hemorrhage and ideally negative GBM titers. Typically for 10-20 days.
Replacement fluid
The replacement fluid is either:
- 5% Albumin
- Used for most cases
- Fresh Frozen Plasma (FFP)
- TTP: FFP contains the deficient ADAMTS 13 protein, the pathologic cause of TTP.
- Coagulation defect or active bleeding.
Plasmapheresis removes clotting factors that are not present in 5% albumin. It takes time for them to be regenerated. If there is a patient with active bleeding (i.e, alveolar hemorrhage) or risk for bleeding (i.e. recent kidney biopsy or significantly low fibrinogen levels) one should use FFP for some of the replacement. 250-500 ml or 1-2 units at the end of the session is appropriate.
Calcium
- If citrate is used as the anticoagulant calcium gluconate 1- 2 grams is typically given to mitigate toxicity.
Adverse effects
These are the potential side effects to be aware of.
- Hypotension: Exposure to the plasmapheresis circuit results in bradykinin generation. Angiotensin system inhibitors (ACE-I) impair the degradation of bradykinin and may increase the risk of hypotension. ACE-I should be held for 24-48 hrs before initiation of plasma exchange.
- Allergic reaction: FFP may result in allergic reactions or transfusion related acute lung injury (TRALI). Pre treatment with antihistamines and/or steroids is often given.
- Citrate toxicity: Manifests as symptomatic hypocalcemia
- Cardiac: Prolonged QT; arrhythmias
- Neurologic: Paresthesia; tetany
- Increased risk with FFP (as FFP contains additional citrate) or renal or hepatic dysfunction (decreased clearance or metabolism).
- Coagulopathy/ Bleeding: Plasmapheresis removes clotting factors. Recovery begins 4 hrs post and is near complete by 48 hours after treatment.
- Hypokalemia/ Hypomagnesemia: 5% albumin contains less potassium and magnesium than plasma and may cause these electrolyte disorders when used as a replacement fluid. In addition citrate may chelate magnesium as it does calcium.
- Medication Clearance:
- Rituximab can be cleared by plasmapheresis. Treatments should be delayed for 24-48 hours after administration
- Other common immunosuppressants (cyclophosphamide, steroids and calcineurin inhibitors) are not significantly affected.
Monitoring:
The following lab studies are often monitored with extended courses of plasma exchange
- CBC
- CMP
- Magnesium
- PT; PTT
- Fibrinogen
- LDH (if TTP)
Indications
Plasmapheresis will rapidly remove existing antibodies, but will not prevent new antibody production. Therefore to adequately treat most conditions concomitant immunosuppression is required
Below are some of the more common indications. This is not a comprehensive list.
Refer to the American Society for Apheresis for a comprehensive list of evidence based guidelines for use of therapeutic plasma exchange in clinical conditions.
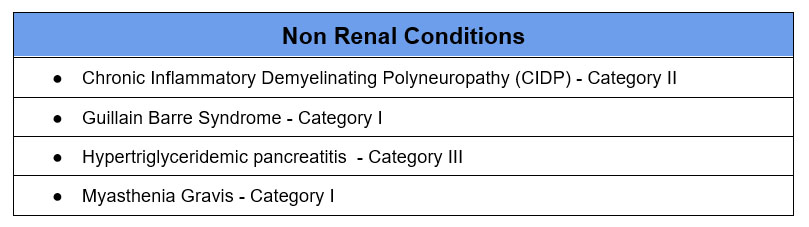
Category I – apheresis is 1st line therapy
Category II – apheresis is 2nd line therapy
Category III – role of apheresis not established, treatment decision should be individualized
Summary
The nephrologist should be familiar with plasmapheresis both because it plays an integral role in treatment of some renal diseases and it is commonly managed by nephrology in the community setting.
For more information check out these excellent reviews.
Therapeutic Plasma Exchange: Core Curriculum 2023 – American Journal of Kidney Diseases